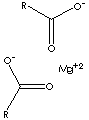|
MAGNESIUM STEARATE | ||
|
PRODUCT IDENTIFICATION |
||
| CAS NO. | 557-04-0 |
|
| EINECS NO. | 209-150-3 | |
| FORMULA | Mg(C17H35CO2)2 | |
| MOL WT. | 591.25 | |
|
HS CODE |
||
| TOXICITY | ||
| SYNONYMS | Octadecanoic Acid, Magnesium Salt; | |
| Magnesium Distearate; Dibasic Magnesium Stearate; Magnesiumdistearat (German); Diestearato de magnesio (Spanish); Distéarate de magnésium (French); | ||
| SMILES | ||
|
CLASSIFICATION |
|
|
|
PHYSICAL AND CHEMICAL PROPERTIES |
||
| PHYSICAL STATE | Fine white to yellow-white powder | |
| MELTING POINT | 150 C | |
| BOILING POINT | ||
| SPECIFIC GRAVITY | 1.03 | |
| SOLUBILITY IN WATER | Insoluble | |
| pH | ||
| VAPOR DENSITY | ||
| AUTOIGNITION |
| |
| NFPA RATINGS | Health: 1; Flammability: 0; Reactivity: 0 | |
REFRACTIVE INDEX |
|
|
| FLASH POINT |
| |
| STABILITY | ||
|
APPLICATIONS |
||
|
Gelling Agent, Stabilizer, Lubricant, Anti-sticking Agent, Sanding Agent, Emulsifier And Plasticizer for Polymer, Paper Pharmaceutical, Rubber, Cosmetic, Paint and Food Industries. |
||
| SALES SPECIFICATION (VEG. GRADE) | ||
|
BIBLIOGRAPHY |
USP 23/ NF 18 | |
|
APPEARANCE |
White powder | |
| IDENTIFICATION |
Pass Test | |
|
Mg CONTENT |
4 - 5% | |
|
STEARIC ACID |
40% min | |
|
HEAVY METALS |
20ppm max | |
|
LEAD |
10ppm max | |
|
CHLORIDES |
0.1% max | |
|
ACIDITY & ALKALINITY |
Pass Test | |
|
SULPHATES |
0.25% max | |
|
FREE FATTY ACID |
0.5% max | |
|
ASH (as MgO) |
7.5% max | |
| TOTAL MICROBIAL COUNT |
1,000/g max | |
| SALMONELLA | Negative | |
| TOTAL MOLDS AND YEASTS |
500/g max | |
| E. COLI | Negative | |
| LOSS ON DRYING |
4.0% max | |
| MELTING POINT |
150 C | |
|
SIEVE ANALYSIS |
99.8% Through 200 mesh | |
| TRANSPORTATION | ||
| PACKING | 20kgs in Bag | |
| HAZARD CLASS | ||
| UN NO. | ||
| GENERAL DESCRIPTION OF METALLIC SALTS OF FATTY ACIDS |
||
| Metallic salts of fatty acids (called soap) are primarily used as cleansing agent (mainly sodium- and potassium-) which their molecules attach readily to both polar molecules (of water) and non-polar molecules (of grease or oil). The long hydrocarbon chains are non-polar (and hydrophobic) repelled by water and the salt end molecules are ionic (and hydrophilic) water soluble. Soaps differ according to the type of fatty acid and length of the carbon chain and according to the alkali employed. Fatty acids with longer chains are insoluble. If sodium hydroxide is used as the alkali, hard soaps are formed; potassium hydroxide yields soft soaps. Soap salts are used as insecticides, herbicides, fungicides and algaecides. The lipophilic carbon chains infiltrate and destroy the lipoprotein matrix of the insect's cell membranes. Food grade soap salts are used also as general purpose food additives. Aluminum, calcium, magnesium, lead, zinc or other metals are used in place of sodium or potassium for soaps to be used in industry. Metallic salts of fatty acids are used as stabilizer and plasticizer in plastic industry as well as in cosmetics. They are used as flatting and sanding agents in lacquers, coatings & inks. They can be applied in tablet manufacturing. They are used as drying lubricants and dusting agents for rubbers. They are used as catalysts in chemical synthesis and emulsifiers for emulsion polymerization of synthetic rubber and resin which can be approved for use in food contact applications. They are used as waterproofing additives and ointments. | ||